Cardiovascular Program Strategy: TAVR for All Moves Ever Closer
Hospitals with small to medium-sized cardiac valve surgery programs need to know that the era of...
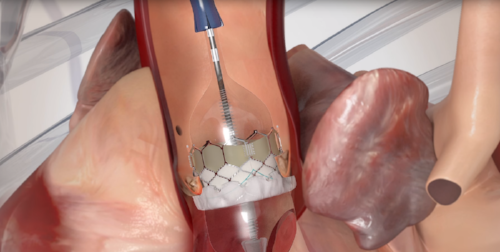
 Two recent developments are important for those hospitals monitoring Transcatheter Aortic Valve Replacement (TAVR) developments.
Two recent developments are important for those hospitals monitoring Transcatheter Aortic Valve Replacement (TAVR) developments.
First, on July 25, 2018 CMS held a meeting of its Medicare Evidence Development and Coverage Advisory Committee (MEDCAC) to discuss procedural volume requirements for TAVR, Percutaneous Coronary Interventions (PCI) and other relevant structural heart disease procedures.[1] Second, prior to the CMS meeting on July 18, 2018, JACC released for pre-publication 2018 AATS/ACC/SCAI/STS Expert Consensus Systems of Care Document: Operator and Institutional Recommendations and Requirements for Transcatheter Aortic Valve Replacement. While both the MEDCAC meeting and the AATS/ACC/SCAI/STS consensus recommendations focused on requirements for new programs and continued certification of existing programs, we would like to focus specifically on issues facing new programs.
Could this be the start of reducing, minimum volume requirements for TAVR reimbursement under Medicare? If that were to happen, how many lower volume (i.e., less than 50 Aortic Valve Replacements per year) cardiac valve surgery programs would enter this market? Although speculative at best, this prospect does initiate an interesting discussion of the pros and cons of entering a growing market and attempting to capture lost referrals for hospitals currently operating what CMS has defined by regulation as “low-volume” valve surgery programs.
Current Required Criteria to Provide TAVR and Receive Reimbursement from CMS
Medicare’s 2012 national coverage decision lists the following criteria to establish a new TAVR program:
As of 2015, 400+ U.S. hospitals performed 54,782 TAVR procedures according to the Annual Report of the TVT Registry.[2] The new expert consensus document states that there are more than 580 active TAVR/SAVR sites with over 100,000 TAVR procedures in the TVT Registry. During 2016, the total number of TAVR and surgical AVRs were about equal. It is predicted that eventually, TAVR will far exceed SAVR volume.
Newly Proposed TAVR Requirements from the Consensus Document[3]
As stated above, JACC recently released a pre-publication version of 2018 AATS/ACC/SCAI/STS Expert Consensus Systems of Care Document: Operator and Institutional Recommendations and Requirements for Transcatheter Aortic Valve Replacement. This new consensus document provides detailed recommendations for institutions and individuals to assess their potential for instituting and/or maintaining a high quality TAVR program (the first update since 2012) and reflects the current evolution in practice and quality benchmarks. At this stage these are multi-societal consensus recommendations and not accepted CMS requirements.
Of particular note are two findings:
In addition, these recommended changes were made to the current recommendations for new programs:
The Natural Progression of Technology
The roll-out of TAVR technology and its application to an increasingly higher-risk target population has been logical and conservative, following the general path that other new technologies have historically taken. This is logically demonstrated in the new AATS/ACC/SCAI/STS consensus documents and its focus on clinical outcomes and performance improvement. We all remember when PCI was introduced in 1977 and how the device evolution (e.g., balloon, bare-metal stent, drug-eluting stent, dissolving stent), patient appropriateness criteria and technological advances evolved to where it is the common go-to procedure today. While there are ACC/AHA guidelines, and in some states, guideline-derived licensing/certification requirements for operator/provider minimum volumes, both individual operators work load requirements and clinical outcomes for PCI are not the issues they once were.
TAVR seems to be following a similar evolutionary and somewhat predictable path. Within this natural evolutionary process, there are always logical concerns of balancing operator skills; minimum proficiency volumes; patient access and selection, appropriateness and risk assessment; clinical and life-style outcomes, and the technological evolution of the procedure, equipment and devices being utilized. Information from the TVT Registry documents clinical and quality-of-life outcomes from TAVR continue to improve over time[4]. If the past is prologue to the future, then TAVR criteria will also eventually be modified, and perhaps inevitably reclassified from “mandatory” to “recommended” status. This could change the market for TAVR providers. The one unanswerable question involves timing – just how long will it take for new guidelines/recommendations to generate new Medicare reimbursement determinations and state-sponsored regulations?
TAVR in Low-Volume Hospitals
If and when TAVR reimbursement criteria is modified, what impact will it have on the number of TAVR providers? Will some procedural volume shift from specialty center to lower volume hospital programs? Is this a positive development? Hospitals that cannot meet the stringent 2012-issued criteria have inevitably lost perhaps fifty percent of their AVR procedures to competitor programs with higher volumes. Some lower volume programs may be able to meet new criteria, fundamentally redistributing overall TAVR volume (and the generally adverse financial consequences) between the 500+ providers now in the market across a greater number of providers. Will the average volume (both TAVR and SAVR) per provider decrease? Unless or until patient selection criteria evolves to include a higher percentage of TAVR candidates (beyond current low and intermediate risk patients), then the size of the market will stay relatively stable and some existing programs will lose volume to low-volume providers entering the market.
Complicating this discussion are research findings based on the TVT Registry that TAVR is associated with better outcomes in high-volume centers. According to Dr. John D. Carroll, “The volume-outcome relationship for TAVR is both statistically significant and clinically important.”[5] CFA believes the interplay between volume standards, operator skill/proficiency and case acuity stratification are major drivers of outcomes. This is, as was the case with low-volume cardiovascular surgery program (LVCSP) outcomes, a multi-factorial issue which requires greater assessment. Today, the clearly documented position in the cardiovascular literature is that cardiac surgery, in the hands of a skilled cardiac surgeon operating in a low volume setting, is as efficacious as those procedures done at larger centers.[6]
Further complicating the discussion was the debate within the MEDCAC meeting, which appeared to be split on the issue of whether procedural volume requirements should be required to begin and maintain TAVR programs.[7]
The Implications of Low-Volume TAVR
If current reimbursement were to improve and/or licensure criteria were to be relaxed, any hospital contemplating entering the TAVR market on a low-volume basis should think long and hard before deciding. Optimizing TAVR programs specifically, and valve programs in general, has been challenging even in high-volume situations. While TAVR reimbursement changed for the better in FY 2015 with dedicated unique MS-DRGs, the cost associated with this procedure typically outstrips the reimbursement.
In a value-driven world, with an increasing focus on episodes of care, this situation is problematic with infrastructure, overhead, and high device costs. If your hospital is losing valve surgery cases, and especially TAVR cases, to higher-volume competitors, entering this market may be a viable alternative. However, there are significant barriers to entry and continuing challenges to going-forward operations and financial viability that make this decision a complex and potentially costly one. Even now, some hospitals have opted not to pursue TAVR business and have successfully optimized their valve programs by providing expert pre-procedural diagnosis and follow-up for TAVR patients while still referring the patient for the procedure to a center of choice. CFA will follow CMS’s reconsideration of volume criteria very carefully. However, a new National Coverage Determination isn’t expected until June 2019.
If you are interested in learning more about low-volume cardiac surgery program strategies, please download our updated and expanded whitepaper (see Low-Volume Cardiac Surgery Programs – Grow, Consolidate or Divest: Self-Preservation Strategies and Excellence Expectations, our newly expanded and updated White Paper). If you are interested in cardiac services strategic development, service expansion and/or other programmatic needs for cardiovascular or other services, please contact CFA at (949) 443-4005 or by e-mail at cfa@charlesfrancassociates.com.
[1] CMS to reconsider TAVR volume requirements, Cardiology Business, accessed online July 02, 2018.
[2] Grover, et al; 2016 Annual Report of the Society of Thoracic Surgeons/American College of Cardiology Transcatheter Valve Therapy Registry, Journal of the American College of Cardiology, December 2016.
[3] Bavaria JE, Tommaso CL, Brindis RG, Carroll JD, Deeb GM, Feldman TE, Gleason TG, Horlick EM, Kavinsky CJ, Kumbhani DJ, Miller DC, Seals AA, Shahian DM, Shemin RJ, Sundt III TM, Thourani VH, 2018 AATS/ACC/SCAI/STS Expert Consensus Systems of Care Document:
Operator and Institutional Recommendations and Requirements for Transcatheter Aortic Valve Replacement, Journal of the American College of Cardiology (2018), doi: 10.1016/j.jacc.2018.07.002.
[4] Grover, F. L., and Holmes, D. R., New Insights from the STS/ACC TVT Registry, Cardiac Interventions Today, Vol. 11, No. 2 March/April 2017.
[5] Carroll JD, et al. Joint ACC/TCT Late-Breaking Clinical Trials. Presented at: American College of Cardiology Scientific Session, April 2-4, 2016; Chicago, IL.
[6] The Society of Thoracic Surgeons Adult Cardiac Surgery Database: 2018 Update on Outcomes and Quality
[7] https://www.cardiovascularbusiness.com/topics/structural-congenital-heart-disease/cms-panel-divided-tavr-volume-thresholds

Hospitals with small to medium-sized cardiac valve surgery programs need to know that the era of...

CMS has been busy. On July 25th they published their Notice of Proposed Rulemaking for Bundled...